Peptides in Orthopaedics: What the 2026 AAOS Review Reveals
The American Academy of Orthopaedic Surgeons just published a comprehensive review on BPC-157, TB-500, and GHK-Cu. Here's what the evidence actually shows about peptides for injury recovery.
The News: Orthopaedic Surgeons Are Taking Peptides Seriously
Something notable happened in early 2026. The American Academy of Orthopaedic Surgeons — not exactly known for rushing into trends — published a comprehensive review on therapeutic peptides in their official journal. The paper, "Therapeutic Peptides in Orthopaedics: Applications, Challenges, and Future Directions," represents a shift in how mainstream medicine views compounds that were, until recently, confined to niche biohacking circles.
The authors — Omar F. Rahman, Steven J. Lee, and William A. Seeds (yes, the Dr. Seeds of SSRP Institute) — aren't just theorizing. They reviewed the mechanistic evidence for BPC-157, TB-500, GHK-Cu, and a roster of growth hormone secretagogues, mapping out exactly how these peptides modulate the molecular signaling networks that drive tissue regeneration.

What the Evidence Actually Shows
Let's be precise about what we know. The review identifies five key peptide classes relevant to orthopaedic care:
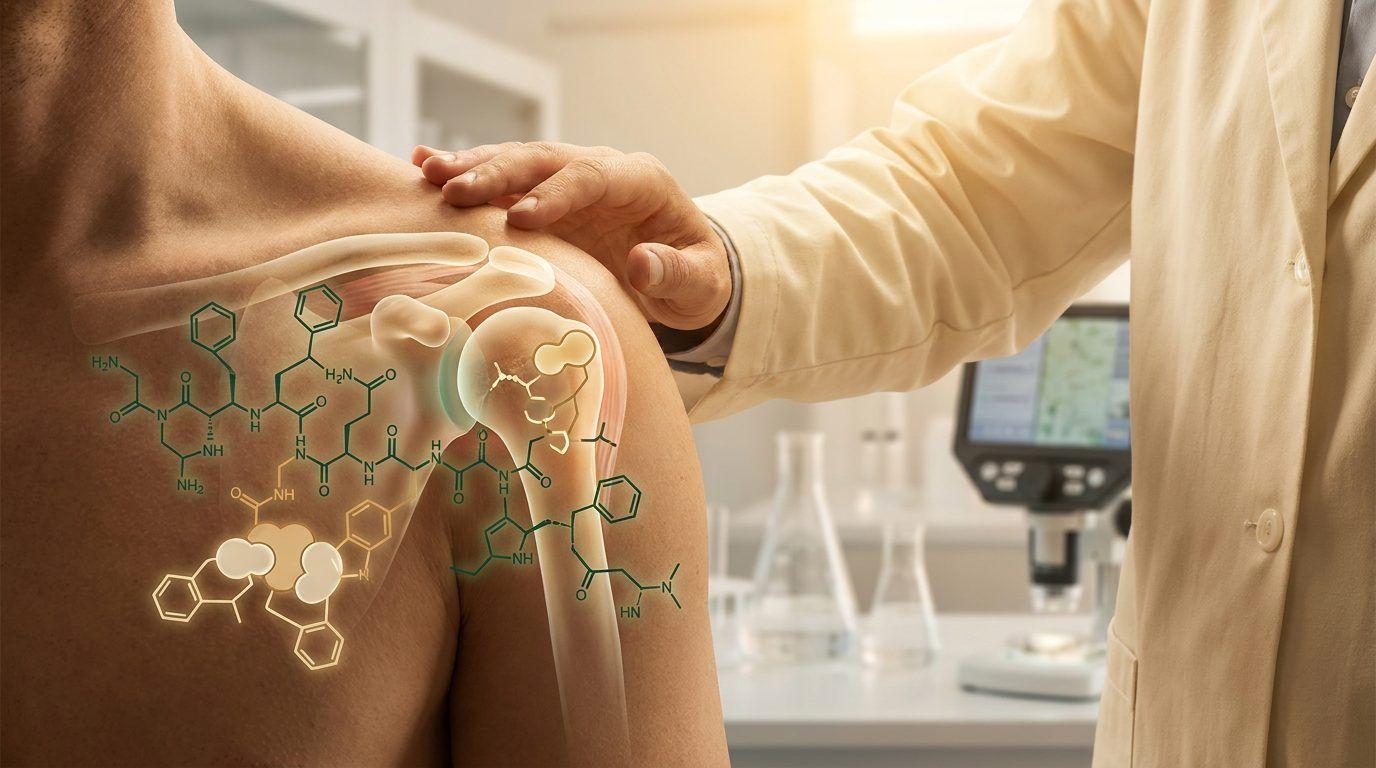
Wound-healing peptides (BPC-157, TB-500, GHK-Cu) work through angiogenesis — the formation of new blood vessels — and extracellular matrix remodeling. BPC-157, a 15-amino acid peptide derived from gastric juice, activates PI3K/Akt and VEGF pathways. TB-500, derived from thymosin beta-4, promotes cell migration and cytoskeleton repair through MAPK and NF-κB pathways. GHK-Cu, the copper-peptide complex, drives collagen turnover via MMP regulation and NRF2 antioxidant pathways.
Growth hormone secretagogues (ipamorelin, CJC-1295, tesamorelin, sermorelin, AOD-9604) stimulate the GH-IGF-1 axis. The review notes that CJC-1295 combined with ipamorelin creates a sustained anabolic environment conducive to muscle repair and chondrocyte activation. AOD-9604 is particularly interesting — it promotes cartilage repair and lipolysis without stimulating IGF-1, working through AMPK activation instead.
Recovery-enhancing agents (epithalon, delta sleep-inducing peptide, pinealon) target circadian rhythms and mitochondrial function. Epithalon, the four-amino acid peptide from the pineal gland, activates SIRT1 and telomerase — relevant not just for sleep, but for cellular senescence in healing tissues.
The Critical Gap: Where Are the Human Trials?
Here's the part that demands honesty: the review authors explicitly state "there is a current lack of clinical trials." Almost all robust evidence for these peptides comes from preclinical models — rat Achilles tendon repairs, mouse muscle crush injuries, in vitro cell culture studies.
This doesn't make the science unimportant. Preclinical studies showing BPC-157 improves collagen alignment in severed rat tendons (published in Journal of Orthopaedic Research, 2003) used rigorous methodology. The mechanisms are real. But translating animal data to human orthopaedic outcomes requires controlled clinical trials that, for the most part, haven't been funded yet.
What This Means for Patients and Practitioners
Dr. Wade McKenna, a Texas-based orthopaedic surgeon, puts it plainly in his clinical blog: "I need to be completely straightforward with you — virtually all of the robust evidence for BPC-157 comes from animal studies, not human clinical trials." He emphasizes proper storage, handling, and sourcing — points the AAOS review also stresses.
The April 2026 FDA announcement — moving 12 peptides (including BPC-157, TB-500, and GHK-Cu) out of Category 2 restrictions — changes the regulatory landscape. These compounds are now on track for formal review beginning July 2026. Licensed compounding pharmacies will be permitted to prepare them again for patients with valid prescriptions under physician supervision.
For orthopaedic applications, this means patients with stubborn Achilles tendinopathies, rotator cuff injuries, or post-surgical recovery needs may have access to peptide adjuncts through legitimate clinical channels — not just research chemical suppliers.
The Oria Take
Peptides like BPC-157 and TB-500 have a genuine mechanistic rationale in orthopaedic healing. The PI3K/Akt, mTOR, and MAPK pathways they influence are the same pathways your body uses to repair tendons, ligaments, and muscle after injury. This isn't hype — it's cellular physiology.
But the absence of human clinical trials in orthopaedics is a gap we need to acknowledge. If you're considering peptide therapy for a musculo-skeletal injury, do it under the care of a trained provider who understands dosing, sourcing, and monitoring. The AAOS review emphasizes that "responsible integration into musculoskeletal care" requires exactly this kind of clinical oversight.
The regulatory winds are shifting. The science is accumulating. And for the first time, mainstream orthopaedic surgery is paying attention. That combination — science plus regulation plus clinical interest — is what moves peptides from biohacker basements to legitimate patient care.
Evidence Grade
**Grade B** — Mechanistic evidence and preclinical studies are strong; human clinical trial data in orthopaedic applications is limited but growing. FDA reclassification (April 2026) signals regulatory recognition of safety profile.
---
*The information provided on this site is intended for educational purposes only and is not a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition or treatment. Never disregard professional medical advice or delay in seeking it because of something you have read here. Peptides discussed are for research purposes unless prescribed by a licensed provider.*
Related Compounds
Build Your Personalised Protocol
Turn your research into a personalised supplementation protocol with the BioStack Generator.